TMS Depression Treatment
TMS Depression treatment at the TMS Institute of Arizona in Scottsdale can be highly effective for the treatment of depression. We use gentle magnetic stimulation to increase brain activities in areas that have become under-active.
This treatment has been FDA-Approved for over 10 years and requires no drugs. It’s completely non-invasive, the magnet used barely touches your body.
Depression is a very common mental disorder that affects more than 300 million people globally and unfortunately, traditional treatment resistance via medication is circa 20%. And even more people struggle with medication side effects.
This makes Transcranial Magnetic Stimulation (TMS) treatment for depression a very valuable treatment. TMS has been extensively researched and is FDA-approved for Major Depressive Disorder.
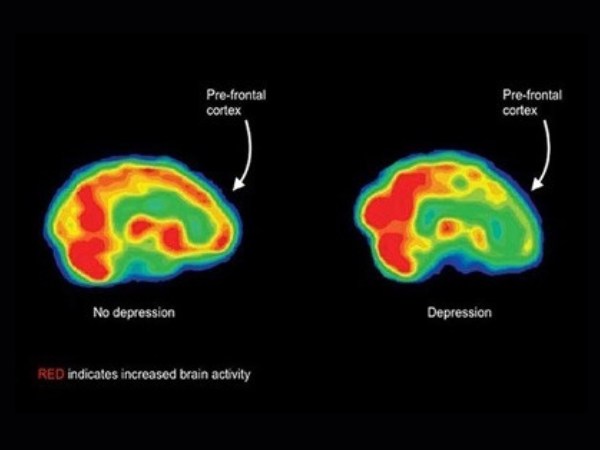
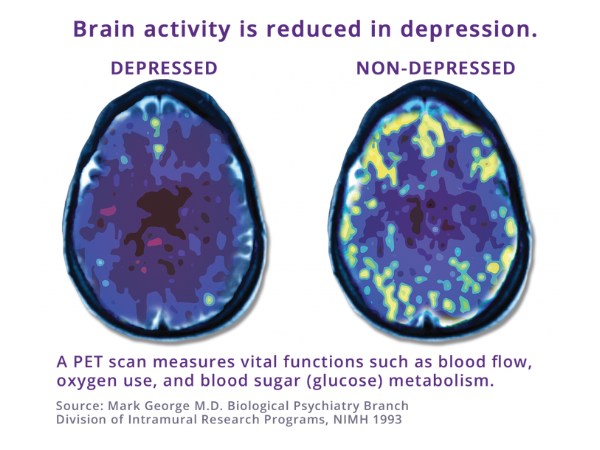
Research into TMS showed that there is asymmetry in the functioning of the frontal lobe of the brain in patients with depression, which makes TMS a great treatment for many patients. TMS Depression treatment improves the functioning in the frontal lobe by increasing activity on the left side of the frontal lobe to reverse the asymmetry found in depressed patients.

Call 480-448-2916 for more information.
Your Depression Doesn’t Have to Win. TMS Can Help.
Depression can often feel like a losing battle. You get some help and feel better for a while. Then something happens, and you’re back where you started — or worse. And despite trying different medications and therapies, the depression isn’t going away. It can be overwhelming.
That’s why we’re so excited about breakthrough depression treatments that are getting some remarkable results: repetitive TMS, Express TMS, and Accelerated TMS. See explanations of each below.
Depression is a complex disorder that can be caused by a variety of factors, including altered neural circuits in the brain. These altered circuits can lead to changes in brain function, which can contribute to symptoms of depression. Various forms of TMS treatments works directly on these altered brainwaves to improve their function.
Your Brain and Depression
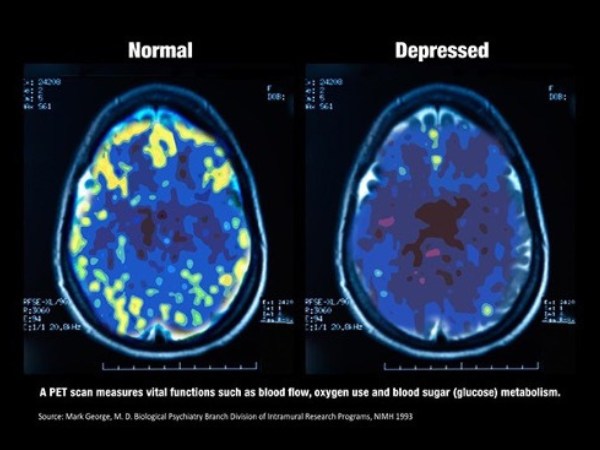
Depression affects everyone differently, as no two brains are alike. Yet, commonalities exist in terms of how depression can affect the brain. Many changes occur in the brain during depression.
- Reduced activity in the prefrontal cortex leads to problems with concentration, motivation, and decision-making.
- Increased activity in the amygdala causes feelings of sadness, hopelessness, and worthlessness.
- Changes in the hippocampus create problems with memory and concentration.
- Changes in the neurotransmitters serotonin and norepinephrine can lead to sadness, fatigue, and appetite changes.
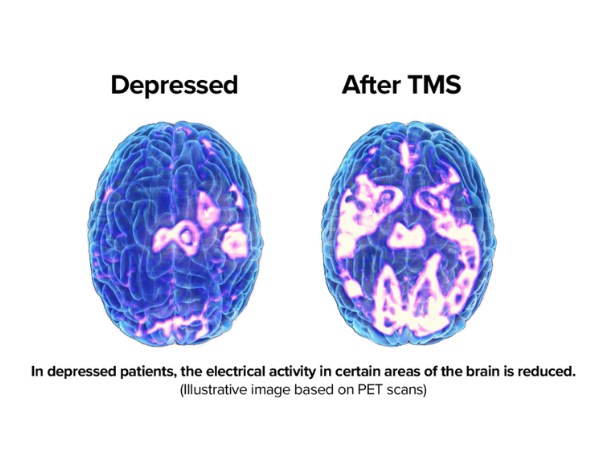
Fortunately, your brain can often repair itself. This concept, known as neuroplasticity, refers to how the brain can change, reorganize, or grow neural networks.
With depression, EEG studies can show an imbalance in brainwave activity between the left and right hemispheres (asymmetry). We then plan a personalized program of TMS to address that asymmetry. As we strengthen your brainwave activity, we can reduce your depression symptoms.
Depression Treatment Studies
There are many peer-reviewed research studies that have shown TMS to be a highly effective treatment for Depression. Here are just some of these with Links to review the full published study.
TMS Should Be Considered as First-Line Treatment For Moderate to Severe Major Depressive Disorder, in Psychiatric News, October 2022. The article’s author, Richard A. Bermudes, reviewed a decade’s worth of studies on the effectiveness of TMS, and wrote this: “As I read the [APA] guidelines recently and considered the number of new outcome studies conducted with TMS, I believe TMS should be considered, in addition to pharmacotherapy and psychotherapy, as a first-line treatment for patients with moderate to severe major depressive disorder.”
A retrospective chart review to assess the impact of alpha- guided transcranial magnetic stimulation on symptoms of PTSD and depression in active-duty special operations service members, 21 June 2024. “This data provides a demonstration of significant reduction in PTSD and depression symptoms and safety with the application of a-rTMS in active-duty special operations military personnel. Expansion of targeted neuromodulation programs could be impactful for military and civilian populations.”
Transcranial magnetic stimulation (TMS) for major depression: a multisite, naturalistic, observational study of quality of life outcome measures in clinical practice, July 2013. This study found: “Conclusion: These data confirm that TMS is effective in the acute treatment of MDD in routine clinical practice settings. This symptom benefit is accompanied by statistically and clinically meaningful improvements in patient-reported QOL [quality of life] and functional status outcomes.”
A naturalistic study of the efficacy and acceptability of rTMS in treating major depressive disorder in Australian youth, June 30, 2024, found the following:
“Results: A 4–7-week course of rTMS significantly reduce symptoms of self-reported depression (42.5% response) and clinician-assessed depression (40.7% response). Both anxiety and stress significantly reduced across the course of rTMS treatment and significant improvements to QoL and self-reported cognition were observed. Reported side effects following rTMS in youth included a mild headache and fatigue.
“Conclusions: The findings of this naturalistic study suggest that an acute course of rTMS provided in private clinical settings is safe and effective – resulting in similar response rates in youth patients as reported in adult patients. In real world practice, rTMS proves to be a well-tolerated and highly effective intervention for MDD in youth, across diverse clinical settings.
“Implications and Contribution: The findings of this naturalistic study suggest that in real world practice, rTMS proves to be a well-tolerated and highly effective intervention for treating depression and anxiety in youth (17–25-year-olds), with additional benefits to cognitive symptoms of depression and overall well-being.”

Use of Transcranial Magnetic Stimulation for Depression, May 2019, stated: “The clinical efficacy of TMS as an antidepressant has been well established. TMS is an innovative and promising treatment modality for patients with TRD [treatment-resistent depression].”
Experimental depression treatment is nearly 80% effective in controlled study, October 28, 2021: “In a double-blind controlled study, high doses of magnetic brain stimulation, given on an accelerated timeline and individually targeted, caused remission in 79% of trial participants with severe depression. “A new type of magnetic brain stimulation brought rapid remission to almost 80% of participants with severe depression in a study conducted at the Stanford University School of Medicine.
Transcranial magnetic stimulation (TMS) for major depression: a multisite, naturalistic, observational study of acute treatment outcomes in clinical practice, Carpenter et al, 2012. This study found as its conclusion: “These data indicate that TMS is an effective treatment for those unable to benefit from initial antidepressant medication.”
Stanford Neuromodulation Therapy (SNT): A Double-Blind Randomized Controlled Trial, Oct 2021: “Major depressive disorder is the leading cause of disability worldwide, and approximately 50% of patients meet criteria for treatment-resistant depression. Repetitive transcranial magnetic stimulation (rTMS), a brain stimulation treatment approved by the U.S. Food and Drug Administration (FDA) for treatment-resistant depression, targets the left dorsolateral prefrontal cortex (DLPFC), a key area in neural circuitry underlying depressive symptoms that has been shown to be hypoactive in major depressive disorder. Contemporary FDA-approved protocols for stimulation of the left DLPFC are limited by the long duration of treatment course (6 weeks) and have been only modestly effective, inducing remission after 4–6 weeks of treatment in ∼17% of patients who have not shown response to three prior antidepressant treatments.”
Efficacy of repetitive transcranial magnetic stimulation in treatment-resistant depression: the evidence thus far, August 2019. This found: “rTMS is rapidly gaining popularity as a treatment modality for depression. There is growing evidence to support its use in patients with depression as a monotherapy or as adjunct with pharmacotherapy. Additionally, rTMS has been found to be safe and effective in pregnant patients and elderly patients…”
Testimonials
Here are some of the things that patients have said after treatment of depression with TMS.
“My adult son who has suffered from both problems his entire life, and has been on medication, realized a positive change after the first visit. We saw dramatic improvement after each visit. This technology is truly a magic bullet for the right candidate. The staff is professional and friendly. The facilities are equally professional.”
“I felt like I was alone even though I was surrounded by love and care. It was just a cloud that would linger over my life every now and again. BTC helped me understand what was going on in my head and after a month of treatment, I was grateful to be DEPRESSION FREE and MEDICATION FREE for the first time in my life.”
“Everything changed when I started treatment. Within two weeks I was sleeping better. I was off all 12 of my meds that I was on when I started treatment, and I was starting to enjoy life again. After the first month I was socializing with people again, I was able to work out for the first time in years, and I started to help my wife with the bills and our budget. A conservative estimate is that I have had a 60 – 70% reduction of the symptoms and have a whole new lease on life. I can focus. I feel good about myself. I feel confident. I laugh again. Most importantly, I can feel the love for and from my family again. I have never met a group of people who truly are solely focused on helping people feel human again. Spend a day here and you will see a miracle.”
What Depression Looks Like
TMS for Depression Options
We believe TMS should be considered a first-line treatment for moderate to severe Major Depressive Disorder (MDD). It can help restore peace of mind and give you the life you want and deserve without the risks and dependency associated with medications. Medications generally result in, at best, a 16% remission rate in depression symptoms, whereas TMS provides up to 80% remission rates. We offer three types of TMS treatments for depression.
TMS (rTMS)
The high-frequency repetitive TMS (rTMS) initial protocol for depression involves high-frequency stimulation.
Covered by most insurances.
Each session initially lasts 19 minutes.
Express TMS®
Intermittent theta burst stimulation (iTBS) is a newer protocol that allows for the delivery of a higher amount of stimulation compared to traditional TMS.
Covered by most insurances.
Requires short 3-minute treatment sessions.
Accelerated TMS
50 treatments in 5 days (10 daily treatments that are 10 minutes each separated by 50-minute breaks).
Not covered by insurance.
We were one of the first centers in the southwest to offer Accelerated TMS.
Call (480) 405-5075 for more information.

For active or retired Military for Depression
TriCare Insurance is Available for active military personnel, veterans, and their families experiencing Depression. The FDA approved the TMS equipment to treat Depression and TriCare approved coverage for Depression in 2016.
Tricare Prime, Select, and TriWest all cover TMS for MDD. In some cases, patients may be experiencing a mixture of MDD, PTSD, and/or Traumatic Brain Injury (TBI) and in these cases, TMS may be an effective treatment. We are in-network with all three and are a part of the VA Community Care Network. Patients with these circumstances would benefit tremendously from TMS.
Depression can occur as a result of things experienced during your service. And it’s very common for other conditions to occur simultaneously, such as PTSD, Traumatic Brain Injury, and Anxiety.
Hear what others have experienced from these TMS for Depression Treatments…
Video: What is TMS Therapy?
Video: Meet Richard, TMS Video Testimonial
Video: Meet Bari, TMS Video Testimonial
Video: TMS Q&A
Video: TMS Therapy and Depression
Video: Oliver’s Story
Talk with our New Patient Coordinator
Our New Patient Coordinator is here to answer all your questions so you can make the most informed decision.
She can explain our treatment protocols, fees, any possible insurance benefits, and more. She can also schedule you for the next step: a consultation with one of our Doctors.
From there, we can determine if you may benefit from TMS treatment, and you can decide if you would like to move forward. We hope you will consider getting more information. You deserve to live a life free from your symptoms — and we want to help you do just that!
Call (480) 405-5075 for more information.
Or fill in the form below and we will contact you.